INTRODUCTION
LESSON PLAN
LECTURE
DIGITAL STORY
ACTIVITY ONE
ACTIVITY TWO
QUIZ
REFERENCES
CONTACT
This lecture is
designed to give you a little bit of background on the theories of
atomic structure that have evolved over the course of history. You will
use
the information in this lecture as a starting point for the research
you will be doing in Activity One. All
of the images below represent a part of the history of atomic
structure. Each image is accompanied by a short audio lecture. A text
script for each lecture can also be found below the images. If you need
to refer back to a specific section of the lecture, you may use the
quick links below to take you to your desired section.
Greeks
James Dalton
Discovery of the Subatomic Particles
The Plum Pudding Model
Rutherford's Gold Foil Experiment
The Bohr Model
The Wave Mechanical Model
Greeks

The ancient philosophers of Greece were the first to start debating the structure of matter. At that time the big battle was between the theory of Aristotle, who said that matter was continuous, and the theory of Democritus, who said that matter was made up of individual particles called atoms. Although his theory was not popular at the time, we all know now that Democritus was correct.
James Dalton

James Dalton gave us the first really coherent theory in atomic structure. Dalton's model of the atom was called the billiard ball model, since he proposed that the atom was an indivisible sphere like the one you see in this image. Dalton's atomic theory had four main postulates. First, he stated that all elements are composed of indivisible particles called atoms. Second, Dalton said that atoms of the same element must be identical, but that atoms of different elements are not identical. Dalton's third postulate stated that atoms can physically mix to form mixtures or chemically combine in whole number ratios to form compounds. Lastly, Dalton said that in a chemical reaction atoms can be separated, joined, or rearranged, but that an atom of one element would not change into an atom of another element.
Discovery of the Subatomic Particles

With the discovery of the subatomic particles, we learned that Dalton's theory was not completely accurate. The discovery of the subatomic particles showed us that the atom was not indivisible, and that it was in fact composed of smaller particles. The first subatomic particle to be discovered was the electron, which was discovered by J.J. Thomson. Thomson discovered the electron using a cathode ray tube like the one you see in this image. Scientists had been using cathode ray tubes to watch fluorescent streams of electricity, like the blue one in this picture, for years with out understanding what electricity actually was. By putting the cathode ray tube into a magnetic field, which is shown in the image by the magnetic deflection yoke, Thomson was able to bend the beam of electricity. By observing the direction of deflection, Thomson was able to determine that electricity was made of small, negatively charged particles called electrons. Since scientists knew the atom was electrically neutral, they set about finding the positive particle that would balance the charge of the electron, the proton. The proton was discovered by Eugene Goldstein, also using the cathode ray tube. The neutron was the last the subatomic particle to be found. James Chadwick discovered the neutron in 1932.
The Plum Pudding Model
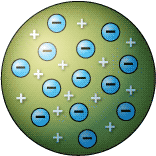
After discovering the electron, J.J. Thomson created a model which he thought represented the structure of the atom. The model was called the Plum Pudding Model because its structure was reminiscent of the desert of the same name. The Plum Pudding Model showed the atom as a positively charged sphere, in which the negatively charged electrons were stuck, like the the chips in a chocolate chip cookie. Thomson's theory was not well received at the time. In order to create an experiment that would provide evidence to support his model, Thomson turned to Ernest Rutherford. The result was Rutherford's famous Gold Foil Experiment.
Rutherford's Gold Foil Experiment

In order to prove Thomson's model of the atom, Rutherford designed an experiment in which alpha particles, which carry a positive charge, would be shot at a thin sheet of gold foil. The theory was that if Thomson's model was correct, the positively charged alpha particles would be repelled from the positive sphere that made up the atom's base structure. Around his experiment Rutherford placed a screen that would detect the repelled alpha particles so that he could see what had happened to the particles. As you can see from the image, what Rutherford and Thomson expected to see was not at all what occurred. Looking at the image, we can see that most of the alpha particles moved straight through the gold foil, while only a small percentage of the particles were deflected. This led Rutherford to make two conclusions about the structure of the atom. First, he concluded that since most of the particles moved straight through the gold foil, the atom was made mostly of empty space. Second, Rutherford concluded that the few alpha particles that were deflected indicated that the atom had a small, dense, positively charged nucleus. Part b of the image shows us what the gold foil looks like on an atomic level. The small spheres you see are not atoms, but the nuclei of the atoms. As you can see, the image accurately represents the idea that the bulk of the atom is comprised of empty space. Rutherford hypothesized that the electrons in the atom were circling the nucleus.
The Bohr Model
Although Rutherford had created a sound atomic model, scientists had just one question for him; why don't the negative electrons get pulled into the positively charged nucleus to which they are attracted? It was Neils Bohr who posed an answer to this question. Bohr proposed a model of the atom in which the electrons were positioned in circular energy levels that orbited the nucleus, as in the image we are looking at now. Bohr stated that each energy level has a defined energy associated with it and that to be positioned in that energy level, an electron must have that specific amount of energy. The energy level closest to the nucleus represented the lowest energy level. As the energy levels grew larger and their distance from the nucleus increased, the energy associated with that level become larger. Bohr said that the electrons could only exist in these levels, although they could move between the levels by absorbing or emitting energy.
The Wave Mechanical Model

Today we know that Bohr's model only explains the behavior of a one electron atom, such as hydrogen. Through time, scientists have developed the wave mechanical model. This model maintains the idea that electrons can only possess certain energies that are associated with principle energy levels in the atom. However, the wave mechanical model does not maintain that the electrons are limited to a specific path as Bohr's model did. Instead, the energy levels in the wave mechanical model are described as regions of probability where you are likely to find an electron that possess a certain energy. In the image we see here, the darker regions of the atom show the areas of high probability for finding electrons associated with a specific energy value.
BACK TO TOP
Greeks
James Dalton
Discovery of the Subatomic Particles
The Plum Pudding Model
Rutherford's Gold Foil Experiment
The Bohr Model
The Wave Mechanical Model
Greeks

The ancient philosophers of Greece were the first to start debating the structure of matter. At that time the big battle was between the theory of Aristotle, who said that matter was continuous, and the theory of Democritus, who said that matter was made up of individual particles called atoms. Although his theory was not popular at the time, we all know now that Democritus was correct.
James Dalton

James Dalton gave us the first really coherent theory in atomic structure. Dalton's model of the atom was called the billiard ball model, since he proposed that the atom was an indivisible sphere like the one you see in this image. Dalton's atomic theory had four main postulates. First, he stated that all elements are composed of indivisible particles called atoms. Second, Dalton said that atoms of the same element must be identical, but that atoms of different elements are not identical. Dalton's third postulate stated that atoms can physically mix to form mixtures or chemically combine in whole number ratios to form compounds. Lastly, Dalton said that in a chemical reaction atoms can be separated, joined, or rearranged, but that an atom of one element would not change into an atom of another element.
Discovery of the Subatomic Particles

With the discovery of the subatomic particles, we learned that Dalton's theory was not completely accurate. The discovery of the subatomic particles showed us that the atom was not indivisible, and that it was in fact composed of smaller particles. The first subatomic particle to be discovered was the electron, which was discovered by J.J. Thomson. Thomson discovered the electron using a cathode ray tube like the one you see in this image. Scientists had been using cathode ray tubes to watch fluorescent streams of electricity, like the blue one in this picture, for years with out understanding what electricity actually was. By putting the cathode ray tube into a magnetic field, which is shown in the image by the magnetic deflection yoke, Thomson was able to bend the beam of electricity. By observing the direction of deflection, Thomson was able to determine that electricity was made of small, negatively charged particles called electrons. Since scientists knew the atom was electrically neutral, they set about finding the positive particle that would balance the charge of the electron, the proton. The proton was discovered by Eugene Goldstein, also using the cathode ray tube. The neutron was the last the subatomic particle to be found. James Chadwick discovered the neutron in 1932.
The Plum Pudding Model

After discovering the electron, J.J. Thomson created a model which he thought represented the structure of the atom. The model was called the Plum Pudding Model because its structure was reminiscent of the desert of the same name. The Plum Pudding Model showed the atom as a positively charged sphere, in which the negatively charged electrons were stuck, like the the chips in a chocolate chip cookie. Thomson's theory was not well received at the time. In order to create an experiment that would provide evidence to support his model, Thomson turned to Ernest Rutherford. The result was Rutherford's famous Gold Foil Experiment.
Rutherford's Gold Foil Experiment

In order to prove Thomson's model of the atom, Rutherford designed an experiment in which alpha particles, which carry a positive charge, would be shot at a thin sheet of gold foil. The theory was that if Thomson's model was correct, the positively charged alpha particles would be repelled from the positive sphere that made up the atom's base structure. Around his experiment Rutherford placed a screen that would detect the repelled alpha particles so that he could see what had happened to the particles. As you can see from the image, what Rutherford and Thomson expected to see was not at all what occurred. Looking at the image, we can see that most of the alpha particles moved straight through the gold foil, while only a small percentage of the particles were deflected. This led Rutherford to make two conclusions about the structure of the atom. First, he concluded that since most of the particles moved straight through the gold foil, the atom was made mostly of empty space. Second, Rutherford concluded that the few alpha particles that were deflected indicated that the atom had a small, dense, positively charged nucleus. Part b of the image shows us what the gold foil looks like on an atomic level. The small spheres you see are not atoms, but the nuclei of the atoms. As you can see, the image accurately represents the idea that the bulk of the atom is comprised of empty space. Rutherford hypothesized that the electrons in the atom were circling the nucleus.
The Bohr Model

Although Rutherford had created a sound atomic model, scientists had just one question for him; why don't the negative electrons get pulled into the positively charged nucleus to which they are attracted? It was Neils Bohr who posed an answer to this question. Bohr proposed a model of the atom in which the electrons were positioned in circular energy levels that orbited the nucleus, as in the image we are looking at now. Bohr stated that each energy level has a defined energy associated with it and that to be positioned in that energy level, an electron must have that specific amount of energy. The energy level closest to the nucleus represented the lowest energy level. As the energy levels grew larger and their distance from the nucleus increased, the energy associated with that level become larger. Bohr said that the electrons could only exist in these levels, although they could move between the levels by absorbing or emitting energy.
The Wave Mechanical Model

Today we know that Bohr's model only explains the behavior of a one electron atom, such as hydrogen. Through time, scientists have developed the wave mechanical model. This model maintains the idea that electrons can only possess certain energies that are associated with principle energy levels in the atom. However, the wave mechanical model does not maintain that the electrons are limited to a specific path as Bohr's model did. Instead, the energy levels in the wave mechanical model are described as regions of probability where you are likely to find an electron that possess a certain energy. In the image we see here, the darker regions of the atom show the areas of high probability for finding electrons associated with a specific energy value.
BACK TO TOP
